Valaciclovir Hcl Overview and Global Sourcing Guide?
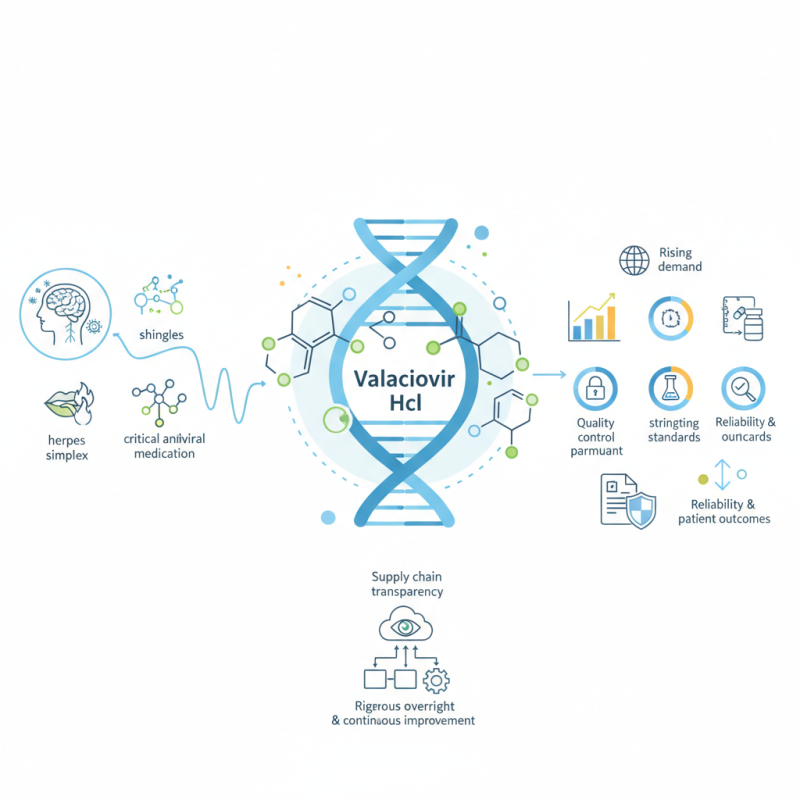
In the ever-evolving pharmaceutical landscape, Valaciclovir Hcl stands out as a critical antiviral medication. Dr. Emily Carter, a leading expert in virology, emphasizes, "Valaciclovir Hcl is a vital resource in managing viral infections effectively." This compound plays a crucial role in treating conditions caused by certain viruses, such as herpes simplex and shingles.
Recent global sourcing trends indicate a rising demand for Valaciclovir Hcl. As manufacturers expand their networks, challenges surface. Quality control remains paramount. The sourcing of raw materials requires vigilance. Each supplier must meet stringent standards. Inconsistent quality can lead to complications in treatment.
Moreover, ensuring the reliability of Valaciclovir Hcl products influences patient outcomes. Stakeholders must prioritize transparency in their supply chains. Failure to address these issues leaves room for uncertainty. Maintaining rigorous oversight is essential to uphold the integrity of this vital medication. The landscape is complex, and continuous improvement is necessary for the future of Valaciclovir Hcl accessibility and efficacy.
Valaciclovir HCl: Chemical Structure and Mechanism of Action
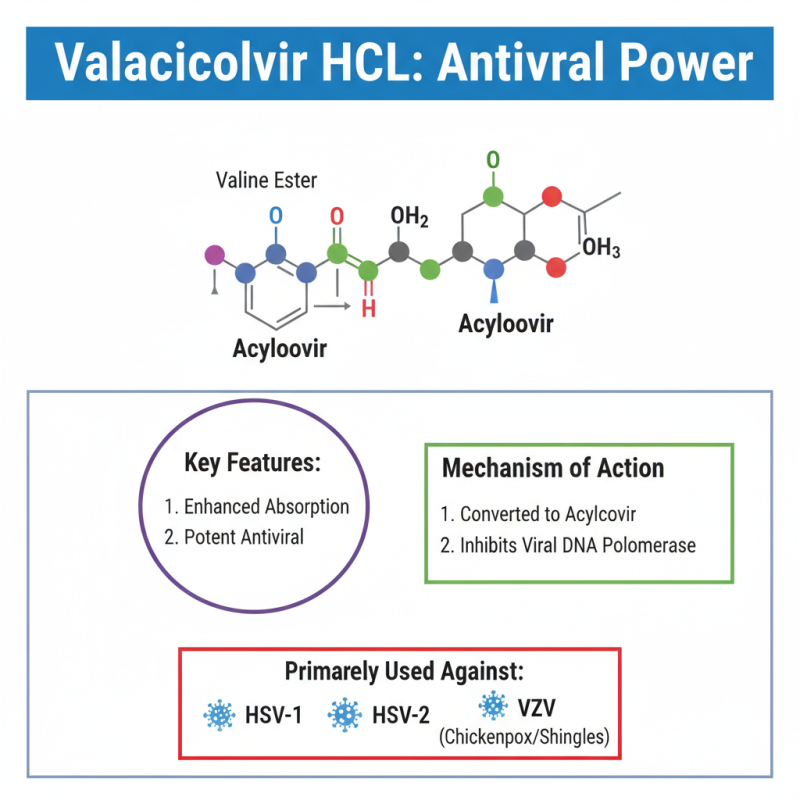
Valaciclovir HCl is an essential antiviral agent. Its chemical structure includes a valine ester of acyclovir. This design enhances its effectiveness in combating viral infections. Valaciclovir is primarily used against herpes viruses. It specifically targets HSV-1, HSV-2, and VZV.
The mechanism of action is quite fascinating. Once ingested, valaciclovir is converted to acyclovir in the body. This conversion allows it to interfere with viral DNA replication. It binds to the viral DNA polymerase, halting the growth of the virus. Cells infected by these viruses are more susceptible to this treatment. However, its effectiveness can vary among individuals, leading to different outcomes.
Research continues to improve formulations and optimize dosages. Insights into resistance patterns are crucial. Making informed decisions about treatment can help. It is important to consult healthcare professionals for tailored advice. Understanding the complexities of valaciclovir is key, yet it is not foolproof. Each case requires careful consideration and evaluation.
Clinical Applications of Valaciclovir in Antiviral Therapy
Valaciclovir is a key antiviral medication used primarily for treating herpes simplex and varicella-zoster viruses. Its clinical applications extend to managing recurrent outbreaks. A 2023 report from the World Health Organization highlights that approximately 500 million people globally are affected by herpes simplex, emphasizing a significant need for effective treatments like valaciclovir.
Clinical trials demonstrate valaciclovir's efficacy in reducing the duration and severity of outbreaks. A study published in the Journal of Antiviral Research states that patients experience a 50% reduction in lesions within 72 hours of initial therapy. Yet, challenges remain, such as the emergence of drug-resistant viral strains. The CDC notes that antiviral resistance can complicate treatment regimens, emphasizing the necessity for careful patient management.
Additionally, valaciclovir's oral form allows for greater accessibility. However, incorrect dosing can lead to inadequate viral suppression. Investigating patient adherence to prescribed therapy is crucial. Studies reveal that nearly 30% of patients fail to complete their regimens as directed. This inconsistency underscores the importance of patient education and monitoring in antiviral therapy. Understanding the broader implications of valaciclovir treatment can improve outcomes and guide future research directions.
Valaciclovir Hcl Overview and Global Sourcing Guide
| Dimension | Details |
|---|---|
| Chemical Name | Valaciclovir Hydrochloride |
| Indications | Treatment of herpes zoster, genital herpes, and cold sores |
| Mechanism of Action | Inhibits viral DNA synthesis |
| Formulation | Tablets available in various strengths |
| Dosage | Typical adult dosage: 500 mg to 1000 mg daily depending on condition |
| Side Effects | Potential effects include nausea, headache, dizziness |
| Global Sourcing Regions | Manufactured in multiple regions including Asia, North America, and Europe |
| Regulatory Status | Approved by FDA and other global regulatory authorities |
Global Market Trends: Valaciclovir Production and Sourcing
The global market for Valaciclovir is evolving rapidly. As a widely used antiviral medication, its production has gained significant traction. Countries with strong pharmaceutical sectors lead in manufacturing. This creates a competitive landscape for sourcing the raw materials.
Production facilities are crucial for meeting demand. High-quality standards are essential in manufacturing Valaciclovir. However, regional disparities in production capabilities can pose risks. Variability in sourcing methods can affect the availability and reliability of this medication.
Understanding market trends is vital for stakeholders. There is a noticeable shift towards sustainable sourcing practices. Companies must navigate regulatory environments carefully. In an ideal scenario, collaboration among producers fosters innovation and quality improvement. Yet, challenges in maintaining consistency still arise. Adapting to new technologies can be a double-edged sword. Balancing efficiency with regulatory compliance requires ongoing reflection and adjustment.
Regulatory Considerations in the Global Distribution of Valaciclovir
Understanding the regulatory landscape for Valaciclovir is crucial for global distribution. Each country has distinct rules regarding pharmaceuticals. Compliance with these regulations is essential for safe and effective distribution. Different jurisdictions impose various testing and approval standards. Keeping abreast of these requirements can be challenging yet necessary.
Documentation plays a significant role in the regulatory process. Properly prepared documents can accelerate approvals. Mistakes or omissions can lead to delays or rejections. Companies must invest time in training staff on regulatory practices. Continuous education helps maintain compliance.
Monitoring regulatory changes is equally important. Regulations can evolve, affecting existing approvals. Companies should have a system for tracking these changes. Regular audits can identify gaps in compliance. This proactive approach can mitigate risks and improve distribution efficiency.
Future Directions in Valaciclovir Research and Development

Recent advancements in valaciclovir research have opened new frontiers in antiviral treatment. As a prodrug of acyclovir, valaciclovir demonstrates enhanced bioavailability. Studies indicate that its half-life can be significantly extended. This characteristic presents unique opportunities for therapeutic applications.
Global data from the pharmaceutical market suggests a growing demand for effective antiviral treatments. The increasing incidence of viral infections, particularly in immunocompromised patients, drives research efforts. For instance, the global antiviral market is projected to reach USD 65 billion by 2028, with valaciclovir playing a pivotal role. Researchers are exploring combinations of valaciclovir with other antiviral agents. These combinations show promise in enhancing efficacy and overcoming resistance.
Despite these advancements, challenges remain. Researchers need to address the side effects associated with long-term use. Moreover, the emergence of resistant viral strains complicates treatment protocols. Continuous monitoring and innovative approaches are essential for overcoming these hurdles. Future studies should focus on optimizing dosing regimens and improving patient compliance.
Related Posts
-

Ensuring Compliance: Industry Standards for the Production of Best Valaciclovir HCl in Global Markets
-
Exploring the Benefits and Uses of Valaciclovir Hcl in Managing Viral Infections
-

Top 5 Benefits of Valaciclovir Hcl You Should Know?
-

How to Use Valaciclovir Hcl Effectively for Treatment?
-

The Ultimate Checklist for Sourcing the Best Valaciclovir Hcl Globally
-

The Future of Valaciclovir Hcl in Antiviral Treatment Innovations





